Shares of BioNTech (NASDAQ: BNTX) were sent into a nosedive on Friday, after a whistleblower report made scathing revelations surrounding trials of the Pfizer-BioNTech Covid-19 vaccine, of which the German company helped develop.
BioNTech plunged by as much as 20% on Friday to around $216 at the time of writing, after a whistleblower’s report revealed dramatic details into the trials of the Pfizer Covid-19 vaccine, which was co-developed alongside BioNTech and US company Pfizer. The report, which was published in the British Medical Journal earlier this week, cites revelations of former contractor Brook Jackson, whom allegedly exposes an alarming amount of incompetence in data handling and patient safety during the trial process.
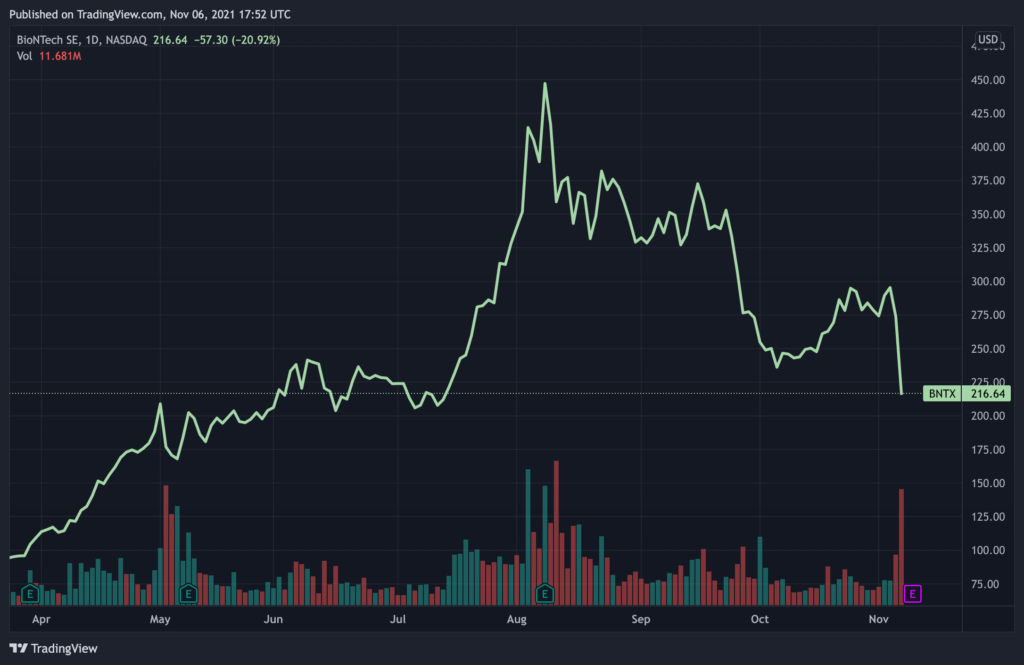
Jackson claims that BioNTech allocated the task of conducting trials to poorly-trained vaccinators, whom failed to investigate adverse side-effects in a timely manner, and even went as far as to falsify certain data. The whistleblower submitted various internal documents to the journal to corroborate her claims.
Some analysts also attribute BioNTech’s dramatic decrease on the Nasdaq to the results of Pfizer’s Covid-19 pill. On Friday, the US company announced its trial data on Paxlovid, its “game-changer” pill, lessens hospitalizations from Covid-19 by 89%. Pfizer’s news also came just as another US pharma company, Merck & Co, revealed that clinical trials for its vaccine reduced the chances of hospitalization and death by 50% in patients with mild to moderate Covid-19 infection.
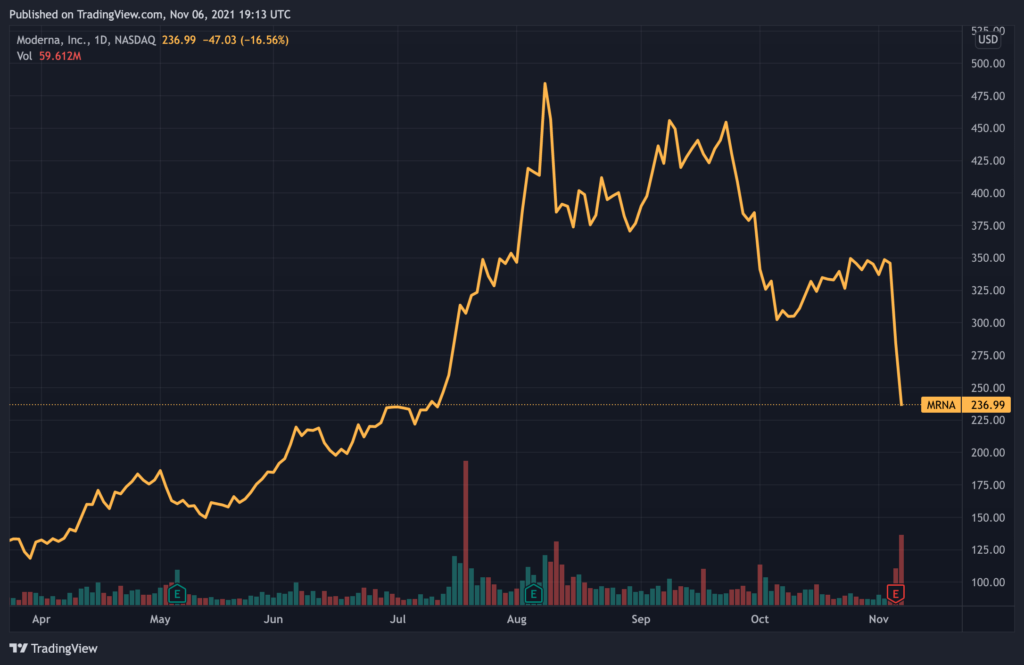
With the emergence of new Covid-19 drugs, investors have begun to anticipate that demand for vaccines would gradually decrease. Moderna, another major vaccine maker, also saw its stock plunge by more than $50 before paring back losses to around $236 at the time of writing.
Information for this briefing was found via Reuters. The author has no securities or affiliations related to this organization. Not a recommendation to buy or sell. Always do additional research and consult a professional before purchasing a security. The author holds no licenses.









