The return volley is almost always a smear job, and it’s easy to see why.
Medical device company Penumbra (NYSE: PEN)‘s November 12th peak valuation of $9.9 billion was achieved in large part through the effective work of internal and external sales desks. That isn’t to say that the company’s reputation as an innovative operator that leads in a high-growth industry isn’t one it’s earned, just that nobody was ever going to be compelled by it as an investment without a sustained and concerted effort to make them care.
The work of short sellers QCM Capital and Marc Cohodes, and to a lesser extent of journalist Roddy Boyd, represents an attempt to destroy the product of that work, and it has been poorly received by the people whose work it is. So they lash out, loading up a mud cannon for a reactionary salvo at the “sleazy short sellers,” who would dare get in the way of the company’s important work marketing “life saving devices.” As they work overtime to make sure they don’t lose control of the broader narrative, this Penumbra team is unmistakably at war.
The Players and the Game
The battle we’re watching play out over Penumbra is not a battle between the longs and the shorts, but rather a battle between the shorts and the company itself, who are being assisted by their allies in sell-side finance. At stake is the institutional capital that is being allocated in the medical devices sector, by way of the decisions of the people in charge of that allocation.
The short syndicate has embarked on a campaign to identify Penumbra as a reckless, dangerous failure that needs new direction and leadership yesterday. Its message to capital is: liquidate this investment before it’s too late. The company’s counter message is that the shorts have over-blown the risks, that the problems are an anomaly, and that their ultimate effect on the company will be minimal.
The state of play
Following Tuesday’s “voluntary” recall of the Jet 7 Xtra Flex catheter (by FDA request), the tone of the company has become more defensive, but its resolve remains strong. A company-hosted conference call that followed the recall featured CEO Adam Elsesser explaining emphatically that the sun doesn’t rise and set on the Jet 7 Xtra Flex. There are other aspiration catheters, and other device verticals, and other places that this great company can take its shareholders in this high-potential medical devices space.
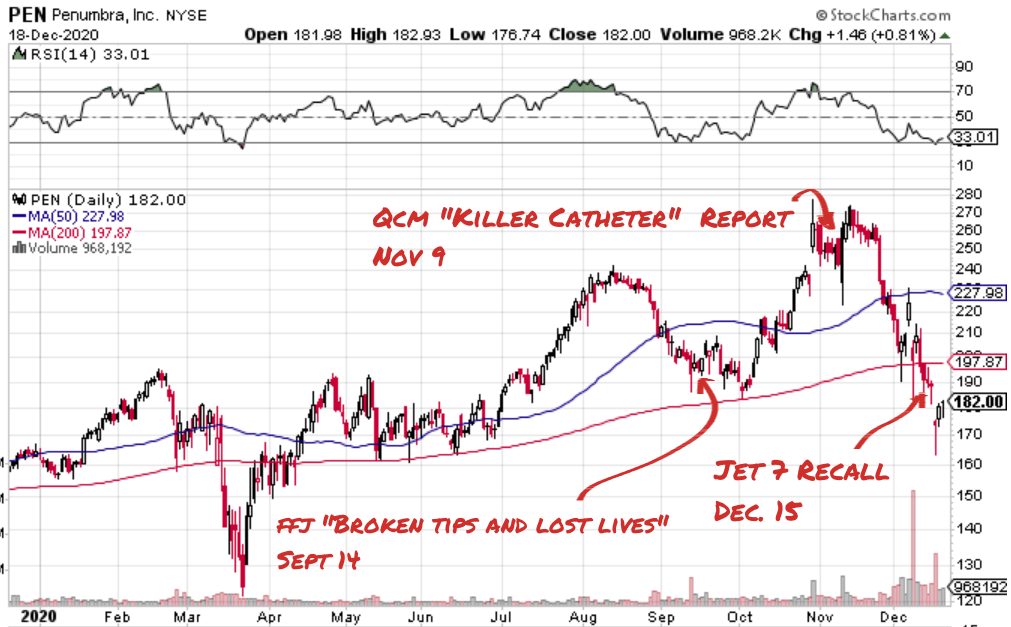
A -7.3% drawdown for Penumbra on the day that followed the recall and its sideways motion ever since suggests, numerologically, that the defensive position is more or less holding. That the collective allocators of the at-risk capital being fought over bought the company line that this catheter is around 7% of total sales, and that some of that is bound to be made up with sales of high-tech stroke catheters in inventory that don’t blow up.
As the warred-over capital’s stewards assess the damage to the $6.3 billion money machine and try to wrap their head around its place on the factory floor of industrialized medicine going forward, QCM is making a fine case that it is unfit for service, and ought to be sold off as scrap. They claim it is under the control of a leadership team that pursues growth over product quality. The drawn-out recall process and the aggressive sales push that followed it seem to bear that out.
The Barbarians at the Gates
So far, the shorts’ diligent work uncovering and publicizing this defect has earned them a -25% slide in Penumbra since the first QCM report dropped, and put them on the business-end of the aforementioned smear. Last week, they seemed a long way from done. Marc Cohodes hosted QCM’s Gabriel Grego on a Periscope live-stream Wednesday in a tone that was downright righteous.
The syndicate is adamant that Penumbra was built on a fraud. The culture that led to the defective product going to market and the subsequent denial and cover up is still part of the company, because the people orchestrating it are still there. It’s rotten to the core, and the problems that landed it there are going to persist and get worse as long as it’s being run by the same team. -25% since the short and -7% since the recall doesn’t seem like it’s nearly enough for these predators now that they’ve got the taste of blood.
We will tear this apart brick by brick until the truth comes out, Elsesser is gone and an Internal Investigation commences @penumbrainc https://t.co/0b2CAtVSWt
— Marc Cohodes (@AlderLaneeggs) December 20, 2020
The initial November 9th report about the deaths in Japan and the United States didn’t hurt Penumbra at first. The stock showed resilience, closing the week out up 8%, suggesting that Penumbra shareholders didn’t find it credible, or believed the company line about the problem being contained, or just didn’t get the memo. PEN’s value saw a steady erosion until December 8th, when it began a sharper slide following QCM’s introduction of a fictional character.
“Dammit, Jim. I’m a DOCTOR!”
It is the very believable allegation of the short-side firm that medical research papers submitted in support of the company’s initial public offering in September of 2015 were authored or co-authored by a person that does not exist. One Dr. Antik Bose, who shares a first initial and last name with former Penumbra Chairman and Chief Medical Officer, and current Penumbra Chief Innovator and Director Dr. Arani Bose, is listed as an author on as many as 44 papers having to do with emergency thrombectomies performed with Penumbra’s developing products. The QCM report links to various social media profiles that describe Dr. Bose as a Turkish hotelier who has done a lot of fine work in the medical field, and is very, very rich.

Everipedia describes him as a billionaire playboy with an expensive estate in upscale Sammamish, WA, and an impressive stable of super cars. The lead video on his youtube account shows “Antik Bose” driving a Rolls Royce Ghost to take viewers on a tour of exotic California properties. The video is an apparently re-purposed and re-labeled version of a video made by luxury Southern California real estate broker Paul Daftarian, who appears to have had his identity cribbed to create a few social media profiles that support the pseudonymous persona.
The “Why?”
QCM unearthed a 2009 instance of Dr. Arani Bose (the one who does exist), apologizing for failing to disclose that he was the Chairman and CMO at Penumbra to medical journal Archives of Neurology (now JAMA Neurology) before submitting an article to the journal, and uses it to support a thesis that the Antik persona was invented to hide the obvious conflicts of interest inherent in a company official being the author on supposedly independent research that details the breakthrough success of its products.
The firm points out that the research being appended to the company’s S1 form at IPO time represents a clear cut case of securities fraud and, if those same papers were used to secure FDA approval for Penumbra products, it would also make a case for the regular kind of fraud.

The company made a tacit admission that the papers were written by Arani Bose in a December 8th press release that describes Antik’s authorship of the papers as a “rumor” in a characteristic shirking of responsibility, and retrenching of its position. It has shown no indication that it is at all worried about material consequences from the misrepresentation, and it’s worth considering whether or not their confidence comes from Antik having long ago hedged against them. QCM makes Antik Bose’s fake profiles full of wealth flexes for a means of gaining friends for the fake profiles, making the seem more real, but that might only be a partial or secondary explanation.
Second cousin to Johnathan Golds
Veterans of the Marijuana stock boom will remember high-profile C21 Investments (CSE: CXXI) shill Johnathan Golds, famous for posting pictures of a blurry Rolex from exotic locales as he assisted a stock promotion that gave the money-losing Nevada cannabis company an out-sized valuation, then disappearing as soon as the sell was on. The lifestyle marketing of financial products is hardly a novelty, but its relevance in this instance is the casting of the avatar as a research scientist who isn’t built to be seen by investors, because the people most likely to look into him are other researchers, publishers of medical journals, and regulators.

Neurothrombectomy is a fairly new field, and Penumbra’s instrumental role in its founding and development made its principles and its early backers a great deal of money. The rapid roll out and development of the first generation of stroke catheters kept the company ahead of its peers until quite recently.
The development and approval of its products and the very adoption of neurothrombectomy as a widespread practice came about after a great deal of work by publishers, independent doctors and researchers, and a lot of regulatory filings. Now, despite work by Medtronic (NYSE: MDT) and Stryker (NYSE: SYK) to crowd them out of it, the company is a leader in the market it created, and its ability to exploit and grow its market share is the only thing that its backers really care about. The business was built at a cost to Penumbra that is small in relation to the value of its equity, but enormous in relation to the earnings of the average research fellow.
Hearts and minds, blood and treasure
So far, none of Bose’s co-authors have come forward to explain how they ended up on a research paper with a fictional character. It’s possible that they wrote the paper with Arani Bose, who was very real, and had the name switched out on them before publication. It’s also possible that they got curious about the switcheroo on Dr. Bose’s first name, looked him up, landed on the profiles full of flashy cars and extravagant homes in Southern California that were a long way beyond their means, and knew damned well that they didn’t just collaborate on a paper with the owner of a Turkish hotel chain, but got the message: there’s lots of money in this for people who play along.
The US Food and Drug Administration, thus far, has been quite accommodating to Penumbra. Both the July Notice to Physicians regarding the Jet 7 and its recall six months later are described as the company’s voluntary actions, done by the agency’s request. A reprimand for submitting research papers by a fake author – assuming the Antik papers were submitted in support of FDA approval – would amount to the FDA admitting to having been fooled in the first place.
Securities regulators and the US DOJ are another matter. The fudged name of an author on papers submitted in support of an S1 would be something it took very seriously in an IPO happening presently. But this one happened five years ago, and the company’s performance since hasn’t given investors any reason to complain. This column had a lot to say in September about US regulators’ lax treatment of JP Morgan Chase following its price fixing in the gold market. The Jay Clayton SEC has not shown any particular willingness to go after bad actors in any meaningful way. Just last week it fined Luckin Coffee (OTCMKTS: LKNCY) $180 million for having outright faked $300 million in revenue. Analysts are calling for Luckin’s stock to double now that it’s out of the woods.
The most recent statements from the short syndicate have included sincere appeals to anyone who might be in a position to come forward as a whistleblower. The emergence of such a figure would surely add gravity to the potential for SEC and FDA investigations, and may even bring about the one event that would represent a decisive victory for the shorts: the resignation or firing of Adam Elsesser and Arani Bose.
Without the executive team, capital allocators aren’t looking at a veteran roster out to prove themselves under pressure. They’re looking at a turnaround story at best, and that’s harder to back. Based on their attitudes so far, this team isn’t likely to concede that hill until the shorts bring a lot more pressure, and they very well may.
Stay tuned.
Information for this briefing was found via Sedar and the companies mentioned. The author has no securities or affiliations related to this organization. Not a recommendation to buy or sell. Always do additional research and consult a professional before purchasing a security. The author holds no licenses.









3 Responses
good! I’m looking forward to seeing your post, will you make it like this again?
This article keeps on saying there were papers written by a fake character, Antik Bose. Name one. One. There are zero. Nada. Nowhere.
Someone created a fake profile on some websites, and claimed they wrote the papers. This strange person also claimed papers by other authors, not just Arani Bose. I can go create fake profiles right now if I want and claim I wrote the Constitution. And these short sellers would say there is fraud because a fake person wrote the Constitution.
The actual papers, which can be found on the real journal sites, are all attibuted to Arani Bose. In all SEC paperwork, Penumbra references papers written by Arani Bose (none by “Antik Bose”). The FDA doesn’t access papers on Researchgate, they go to actual authoritative sources to
Thanks for reading, Robert.
Antik’s profiles full of papers written by Arani have been removed, but survive on the Wayback Machine.
The papers are indeed attributed to A.Bose, including the ones on Research Gate, whose real-life persona was, at the time of their publication, the CMO of the company whose products were central to the paper itself.
It’s the shorts’ allegation that the fake profile was made to duck disclosure obligations. The profiles having been removed since the publication of the QCM report is not consistent with their having been created by some crank for reasons that have nothing to do with Penumbra.
P-10 implementation paper Antik
P-10 implementation paper Arani